The complement inhibitors market is projected to grow, driven by the rising prevalence of complement-mediated diseases, intensified R&D efforts, and biotechnological advancements enabling novel therapeutic development. Increased awareness among clinicians and the approval of new agents such as ULTOMIRIS [AstraZeneca (Alexion Pharmaceuticals)], Sefaxersen (F. Hoffmann-La Roche/Ionis Pharmaceuticals), KP104 (Kira Pharmaceuticals), Ruxoprubart (NM8074) (NovelMed Therapeutics), Riliprubart (Sanofi), AMY-101 (Amyndas Pharmaceuticals), and others are broadening treatment options, collectively accelerating market expansion.
New York, USA, April 30, 2026 (GLOBE NEWSWIRE) — Complement Inhibitors Market Outlook Remains Strong During the Forecast Period (2026–2036) with Increasing Adoption of C5 and C3 Targeted Drugs | DelveInsight
The complement inhibitors market is projected to grow, driven by the rising prevalence of complement-mediated diseases, intensified R&D efforts, and biotechnological advancements enabling novel therapeutic development. Increased awareness among clinicians and the approval of new agents such as ULTOMIRIS [AstraZeneca (Alexion Pharmaceuticals)], Sefaxersen (F. Hoffmann-La Roche/Ionis Pharmaceuticals), KP104 (Kira Pharmaceuticals), Ruxoprubart (NM8074) (NovelMed Therapeutics), Riliprubart (Sanofi), AMY-101 (Amyndas Pharmaceuticals), and others are broadening treatment options, collectively accelerating market expansion.
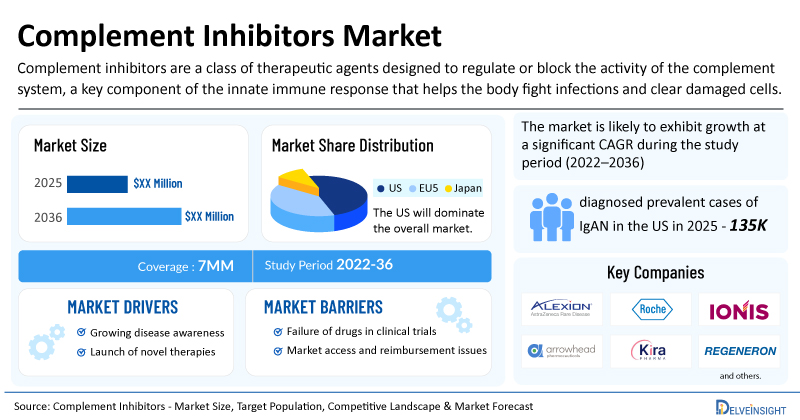
Recently published Complement Inhibitors Market Size, Target Population, Competitive Landscape & Market Forecast report includes a comprehensive understanding of current treatment practices, addressable patient population, which includes top indications such as IgA Nephropathy (IgAN), Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), Paroxysmal Nocturnal Hemoglobinuria (PNH), Immune Complex Membranoproliferative Glomerulonephritis (IC-MPGN), Atypical Hemolytic Uremic Syndrome (aHUS), Complement 3 Glomerulopathy (C3G), and others. The selected indications are based on approved therapies and ongoing pipeline activity. The report also provides insights into the emerging complement inhibitors, market share of individual therapies, and current and forecasted market size from 2022 to 2036, segmented into leading markets (the US, EU4, UK, and Japan).
Key Takeaways from the Complement Inhibitors Market Report
- The total complement inhibitors treatment market size is expected to grow positively by 2036 in the leading markets.
- The United States accounts for the largest market size of complement inhibitors, in comparison to EU4 (Germany, Italy, France, and Spain), the UK, and Japan.
- The report provides the total potential number of patients in the indications, such as IgA Nephropathy (IgAN), Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), Paroxysmal Nocturnal Hemoglobinuria (PNH), Immune Complex Membranoproliferative Glomerulonephritis (IC-MPGN), Atypical Hemolytic Uremic Syndrome (aHUS), Complement 3 Glomerulopathy (C3G), and others.
- IgAN reported approximately 135,000 diagnosed prevalent cases in the US. Improved diagnostic practices, greater disease awareness, and wider use of kidney biopsies is expected to drive the growing prevalence of IgAN.
- Leading Complement inhibitor companies, such as AstraZeneca (Alexion Pharmaceuticals), F. Hoffmann-La Roche, Ionis Pharmaceuticals, Arrowhead Pharmaceuticals, Kira Pharmaceuticals, Regeneron Pharmaceuticals, NovelMed Therapeutics, Sanofi, Amyndas Pharmaceuticals, and others, are actively working on innovative complement inhibitors.
- Some of the key complement inhibitors in clinical trials include ULTOMIRIS (ravulizumab), Sefaxersen (RG6299; IONIS-FB-LRx), ARO-C3, KP104, Pozelimab + Cemdisiran, Ruxoprubart (NM8074), Riliprubart, AMY-101, and others. These novel complement inhibitors are anticipated to enter the complement inhibitors market in the forecast period and are expected to change the market.
Discover C5 inhibitor market growth projections @ https://www.delveinsight.com/sample-request/complement-inhibitors-market-forecast
Key Factors Driving the Complement Inhibitors Market
- Rising Prevalence of Complement-Mediated Disorders: Growing diagnosis rates of diseases such as paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), generalized myasthenia gravis (gMG), neuromyelitis optica spectrum disorder (NMOSD), and geographic atrophy are significantly increasing demand for complement-targeted treatments.
- Expanding Approved Indications: Initially focused on ultra-rare hematologic diseases, complement inhibitors are now gaining approvals across neurology, nephrology, and ophthalmology, broadening the addressable patient population and commercial potential.
- Strong Commercial Success of First-Generation Products: The success of therapies such as Eculizumab and Ravulizumab has validated the complement pathway as an attractive therapeutic target, encouraging further investment and pipeline expansion.
- Launch of Emerging Therapies: The dynamics of the complement inhibitors are expected to change in the coming years due to the launch of emerging therapies such as ULTOMIRIS (ravulizumab) [AstraZeneca (Alexion Pharmaceuticals)], Sefaxersen (RG6299; IONIS-FB-LRx) (F. Hoffmann-La Roche/Ionis Pharmaceuticals), ARO-C3 (Arrowhead Pharmaceuticals), KP104 (Kira Pharmaceuticals), Pozelimab + Cemdisiran (Regeneron Pharmaceuticals), Ruxoprubart (NM8074) (NovelMed Therapeutics), Riliprubart (Sanofi), AMY-101 (Amyndas Pharmaceuticals), and others.
Ramandeep Singh, Senior Consultant of Forecasting at DelveInsight, commented that as newer generations of complement inhibitors target different aspects of the complement cascade (e.g., proximal vs. terminal inhibition), the potential to develop more effective and safer treatments is growing.
Complement Inhibitors Market Analysis
- Complement inhibitor therapies are developed to control overactivation of the complement system, which plays a major role in several immune-mediated disorders.
- By precisely blocking specific elements of the complement cascade, these treatments help reduce inflammation, prevent tissue injury, and ease disease symptoms, providing a more focused strategy for disease management.
- Within this class, C5 inhibitors such as SOLIRIS and ULTOMIRIS remain the most widely established therapies.
- SOLIRIS was the first approved agent and works by blocking C5 activation, which prevents membrane attack complex (MAC) formation and reduces hemolysis and thrombotic complications in diseases such as Paroxysmal Nocturnal Hemoglobinuria and Atypical Hemolytic Uremic Syndrome.
- ULTOMIRIS offers similar efficacy with longer dosing intervals, improving convenience and supporting better adherence.
- SYFOVRE is approved for the treatment of Geographic Atrophy, while EMPAVELI is indicated for Paroxysmal Nocturnal Hemoglobinuria. Both therapies act by targeting abnormal complement activity that contributes to disease progression.
- Although treatment costs remain substantial, SOLIRIS and ULTOMIRIS continue to hold a strong presence in the PNH market because of their proven clinical benefit. Patient assistance initiatives such as Alexion OneSource also help expand treatment access.
- Ongoing research is increasingly directed toward inhibitors of upstream complement targets such as C1 and C3, to treat a broader range of conditions, including Autoimmune Hemolytic Anemia and neuroinflammatory diseases.
- These next-generation therapies highlight a move toward precision modulation of the complement pathway, creating new possibilities for patients who previously had limited therapeutic choices.
Learn more about the global complement inhibitors CAGR outlook @ Complement Inhibitors Analysis
Complement Inhibitors Competitive Landscape
Some of the complement inhibitors under development include ULTOMIRIS (ravulizumab) [AstraZeneca (Alexion Pharmaceuticals)], Sefaxersen (RG6299; IONIS-FB-LRx) (F. Hoffmann-La Roche/Ionis Pharmaceuticals), ARO-C3 (Arrowhead Pharmaceuticals), KP104 (Kira Pharmaceuticals), Pozelimab + Cemdisiran (Regeneron Pharmaceuticals), Ruxoprubart (NM8074) (NovelMed Therapeutics), Riliprubart (Sanofi), AMY-101 (Amyndas Pharmaceuticals), and others.
AstraZeneca’s ULTOMIRIS (ravulizumab) is an extended-duration monoclonal antibody designed to block the C5 protein within the terminal complement cascade, enabling rapid, complete, and durable complement inhibition. By acting on this pathway, ULTOMIRIS helps reduce excessive immune system activation that may otherwise harm healthy cells. The therapy is currently under investigation in the Phase III ICAN clinical trial for the treatment of IgAN. It is also covered by patent protection in key global markets, with expected expiry in the United States in 2035 (extendable to 2038), in the European Union through 2035 (with extensions to 2038–2040), and in Japan until 2038.
Roche and Ionis Pharmaceuticals are jointly developing Sefaxersen (RG6299; IONIS-FB-LRx), an antisense oligonucleotide designed to reduce complement factor B gene expression by targeting factor B mRNA. It is the first ASO aimed at selective complement pathway suppression in IgAN. In July 2022, Roche exercised its option to license IONIS-FB-LRx following positive Phase II results. Roche is now responsible for global development, regulatory, and commercialization activities and associated costs, excluding the open-label Phase II study in patients with IgAN, which remains under Ionis’ conduct and funding.
Omeros’ Zaltenibart (OMS906) is a humanized monoclonal antibody designed to inhibit mannan-binding lectin-associated serine protease-3 (MASP-3), a primary activator of the complement alternative pathway. This pathway plays a role in numerous diseases, including conditions addressed by approved and emerging alternative pathway inhibitors. Unlike C3 and C5 inhibitors, blocking MASP-3 does not disrupt the lytic function of the classical complement pathway, which remains essential for the body’s adaptive immune defense against infections.
In a single-ascending-dose Phase I trial involving healthy volunteers, the therapy demonstrated good tolerability with no notable safety concerns. The FDA has granted Orphan Drug Designation (ODD) to Zaltenibart (OMS906) for the treatment of Paroxysmal Nocturnal Hemoglobinuria. At present, two open-label, multicenter clinical studies are underway to assess the drug in adult patients with Paroxysmal Nocturnal Hemoglobinuria.
NovelMed Therapeutics’ Ruxoprubart (NM8074) is an anti-Bb monoclonal antibody designed to selectively inhibit the alternative complement pathway while preserving the classical pathway, which remains essential for infection clearance. In a Phase I study involving 40 healthy volunteers, the therapy demonstrated a favorable safety profile, was well tolerated at all tested dose levels, and achieved complete inhibition of the alternative pathway, with longer inhibition durations observed at higher doses.
The drug is currently being evaluated in a Phase II trial in treatment-naive paroxysmal nocturnal hemoglobinuria (PNH) patients, where early findings appear encouraging. Beyond PNH, Ruxoprubart may also have therapeutic potential in other diseases driven by dysregulation of the alternative complement pathway. The U.S. Food and Drug Administration has authorized Phase Ib/II clinical studies for PNH, C3 Glomerulopathy (C3G), atypical hemolytic uremic syndrome (aHUS), and most recently, ANCA-associated vasculitis (AAV).
Sanofi’s investigational humanized IgG4 monoclonal antibody, Riliprubart, is designed to selectively block activated C1s, a serine protease involved in the classical complement pathway. The therapy is currently in Phase III clinical development for treating Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), particularly in patients who are refractory to standard of care (SoC) treatments or receiving Intravenous Immunoglobulin (IVIg) therapy.
The anticipated launch of these emerging therapies are poised to transform the complement inhibitors market landscape in the coming years. As these cutting-edge therapies continue to mature and gain regulatory approval, they are expected to reshape the complement inhibitors market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about who are the leading companies in complement inhibitors, visit @ Complement Inhibitors Treatment
Recent Developments in the Complement Inhibitors Market
- In April 2026, AstraZeneca reported that its Phase III trial of ULTOMIRIS (ravulizumab) in adults with immunoglobulin A nephropathy (IgAN) successfully achieved its primary endpoint, paving the way for regulatory submissions for the long-acting C5 complement inhibitor in this disease.
- In March 2026, NovelMed Therapeutics Inc. announced two significant milestones in the clinical advancement of Ruxoprubart (NM8074) for the treatment of Paroxysmal Nocturnal Hemoglobinuria (PNH).
- In January 2026, Sobi announced that the European Commission (EC) had approved pegcetacoplan for the treatment of adult and adolescent patients aged 12 to 17 years with C3G in combination with a renin-angiotensin system (RAS) inhibitor, unless RAS inhibitor treatment is not tolerated or contraindicated.
- In November 2025, Otsuka received FDA accelerated approval for VOYXACT (sibeprenlimab-szsi) for the reduction of proteinuria in adults with primary IgAN at risk for disease progression.
- In October 2025, Novartis’ FABHALTA (iptacopan) met the Phase III primary endpoint and slowed kidney function decline in patients with IgAN.
What are Complement Inhibitors?
Complement inhibitors are a class of therapeutic agents designed to regulate or block the activity of the complement system, a key component of the innate immune response that helps the body fight infections and clear damaged cells. While essential for normal immunity, uncontrolled or excessive complement activation can lead to inflammation, tissue injury, and the progression of various autoimmune, hematologic, renal, and neurological disorders. Complement inhibitors work by targeting specific proteins within the complement cascade, such as C1s, C3, C5, factor B, or factor D, to prevent downstream inflammatory responses and cell destruction. These therapies have emerged as an important treatment approach for diseases such as paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), myasthenia gravis, geographic atrophy, and other complement-mediated conditions.
Complement Inhibitors Epidemiology Segmentation
The complement inhibitors market report is a comprehensive and specialized analysis, offering in-depth epidemiological insights for the study period 2022–2036 across the leading markets. The complement inhibitor target patient pool is segmented into:
- Total Diagnosed Prevalent Cases of Selected Indication with Complement Inhibitors
- Total Eligible Patient Pool for Complement Inhibitors Targeting Therapies in Selected Indications
- Total Treated Cases for Complement Inhibitors Targeting Therapies in Selected Indications
| Complement Inhibitors Report Metrics | Details |
| Study Period | 2022–2036 |
| Complement Inhibitors Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Key Indications Covered in the Report | Paroxysmal Nocturnal Hemoglobinuria, Non-small Cell Lung Cancer, Chronic Inflammatory Demyelinating Polyneuropathy, Complement 3 Glomerulopathy, and others |
| Key Complement Inhibitor Companies | AstraZeneca (Alexion Pharmaceuticals), F. Hoffmann-La Roche, Ionis Pharmaceuticals, Arrowhead Pharmaceuticals, Kira Pharmaceuticals, Regeneron Pharmaceuticals, NovelMed Therapeutics, Sanofi, Amyndas Pharmaceuticals, Novartis, Apellis, Sobi, Astellas, and others |
| Key Complement Inhibitors | ULTOMIRIS (ravulizumab), Sefaxersen (RG6299; IONIS-FB-LRx), ARO-C3, KP104, Pozelimab + Cemdisiran, Ruxoprubart (NM8074), Riliprubart, AMY-101, SOLIRIS, FABHALTA, EMPAVELI/ASPAVELI, IZERVAY, and others |
Scope of the Complement Inhibitors Market Report
- Complement Inhibitors Therapeutic Assessment: Complement Inhibitors current marketed and emerging therapies
- Complement Inhibitors Market Dynamics: Conjoint Analysis of Emerging Complement Inhibitors Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Unmet Needs, KOL’s views, Analyst’s views, Complement Inhibitors Market Access and Reimbursement
Discover more about upcoming complement inhibitors in 2026 @ Complement Inhibitors Clinical Trials
Table of Contents
| 1 | Complement Inhibitor Market Key Insights |
| 2 | Complement Inhibitor Market Report Introduction |
| 3 | Key Highlights from Complement Inhibitor Market Report |
| 4 | Executive Summary of Complement Inhibitors |
| 5 | Key Events |
| 6 | Epidemiology and Market Forecast Methodology |
| 7 | Complement Inhibitors Market Overview at a Glance in the 7MM |
| 7.1 | Market Share (%) Distribution by Therapies in 2025 |
| 7.2 | Market Share (%) Distribution by Therapies in 2036 |
| 8 | Disease Background and Overview |
| 8.1 | Introduction |
| 8.2 | Evolution of Complement Inhibitors |
| 8.3 | Treatment |
| 9 | Target Complement Inhibitor Patient Pool |
| 9.1 | Key Findings |
| 9.2 | Assumptions and Rationale: 7MM |
| 9.2.1 | Total Prevalent Cases in Selected Indications for Complement Inhibitors in the 7MM |
| 9.2.2 | Total Eligible Patient Pool for Complement Inhibitors in Selected Indications in the 7MM |
| 9.2.3 | Total Treated Cases in Selected Indications for Complement Inhibitors in the 7MM |
| 10 | Marketed Complement Inhibitors |
| 10.1 | Key Cross Competition |
| 10.2 | FABHALTA (iptacopan): Novartis |
| 10.2.1 | Drug Description |
| 10.2.2 | Regulatory Milestones |
| 10.2.3 | Other Developmental Activities |
| 10.2.4 | Ongoing Clinical Developmental Activities |
| 10.2.5 | Safety and Efficacy |
| 10.2.6 | Analyst Views |
| 10.3 | EMPAVELI (pegcetacoplan): Apellis Pharmaceuticals |
| 10.4 | IZERVAY (avacincaptad pegol intravitreal solution): Astellas Pharma |
| *List to be continued in the full report | |
| 11 | Emerging Complement Inhibitors |
| 11.1 | Key Competitors |
| 11.2 | Sefaxersen: Roche/Ionis Pharmaceuticals |
| 11.2.1 | Drug Description |
| 11.2.2 | Other Developmental Activity |
| 11.2.3 | Clinical Trial Information |
| 11.2.4 | Safety and Efficacy |
| 11.2.5 | Analyst Views |
| 11.3 | Riliprubart: Sanofi |
| 11.4 | Ruxoprubart (NM8074): NovelMed Therapeutics |
| *List to be continued in the full report | |
| 12 | Complement Inhibitors Market: 7MM analysis |
| 12.1 | Key Findings |
| 12.2 | Complement Inhibitor Market Outlook |
| 12.3 | Conjoint Analysis |
| 12.4 | Key Complement Inhibitor Market Forecast Assumptions |
| 12.5 | Complement Inhibitor Market Size by Indications in the 7MM |
| 12.6 | Complement Inhibitor Market Size by Therapies in the 7MM |
| 12.7 | United States Complement Inhibitor Market |
| 12.7.1 | Market Size by Indications in the US |
| 12.7.2 | Market Size by Therapies in the US |
| 12.8 | EU4 and the UK Complement Inhibitor Market |
| 12.9 | Japan Complement Inhibitor Market |
| 13 | Complement Inhibitor Market Unmet Needs |
| 14 | Complement Inhibitor Market SWOT Analysis |
| 15 | KOL Views on Complement Inhibitors |
| 16 | Complement Inhibitor Market Access and Reimbursement |
| 17 | Bibliography |
| 18 | Complement Inhibitor Market Report Methodology |
Related Reports
IgA Nephropathy Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key gMG companies, including Novartis, Vertex Pharmaceuticals, Biogen, Vera Therapeutics, AstraZeneca (Alexion Pharmaceuticals), F. Hoffmann-La Roche, Ionis Pharmaceuticals, Takeda Pharmaceutical, Arrowhead Pharmaceuticals, NovelMed, Walden Biosciences, Kira Pharmaceuticals, Purespring Therapeutics, Biohaven Therapeutics, and others.
Complement 3 Glomerulopathy Market
Complement 3 Glomerulopathy Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key C3G companies, including Novartis, Apellis Pharmaceuticals, Sobi, Kira Pharmaceuticals, Novo Nordisk, Omeros Corporation, Arrowhead Pharmaceuticals, and others.
Paroxysmal Nocturnal Hemoglobinuria Market
Paroxysmal Nocturnal Hemoglobinuria Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key PNH companies, including Hoffmann-La Roche, Alexion Pharmaceuticals, Novartis, Regeneron Pharmaceuticals, BioCryst Pharmaceuticals, and others.
Chronic Inflammatory Demyelinating Polyneuropathy Market
Chronic Inflammatory Demyelinating Polyneuropathy Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key CIDP companies, including Sanofi, Janssen Research and Development, LLC, Immunovant Sciences GmbH/HanAll Pharma, Roivant Sciences, Inc., and others.
Immune Complex Membranoproliferative Glomerulonephritis Market
Immune Complex Membranoproliferative Glomerulonephritis Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key IC-MPGN companies, including Novartis Pharmaceuticals, Apellis Pharmaceuticals, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur [email protected] +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaChron.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaChron.com takes no editorial responsibility for the same.