The cervical cancer diagnostics market is witnessing significant growth due to the rising prevalence of cervical cancer and increasing awareness regarding early screening and diagnosis. Growing adoption of advanced diagnostic technologies such as HPV testing, liquid-based cytology, and AI-powered screening tools is further accelerating market expansion. Government initiatives and large-scale screening programs, especially in emerging economies, are improving access to cervical cancer diagnostics.
New York, USA, May 14, 2026 (GLOBE NEWSWIRE) — Global Cervical Cancer Diagnostics Market to Exhibit Growth at a CAGR of ~5% by 2034 | DelveInsight
The cervical cancer diagnostics market is witnessing significant growth due to the rising prevalence of cervical cancer and increasing awareness regarding early screening and diagnosis. Growing adoption of advanced diagnostic technologies such as HPV testing, liquid-based cytology, and AI-powered screening tools is further accelerating market expansion. Government initiatives and large-scale screening programs, especially in emerging economies, are improving access to cervical cancer diagnostics.
DelveInsight’s Cervical Cancer Diagnostics Market Insights report provides the current and forecast market analysis, individual leading cervical cancer diagnostics companies’ market shares, challenges, cervical cancer diagnostics market drivers, barriers, trends, and key cervical cancer diagnostics companies in the market.
Cervical Cancer Diagnostics Market Summary
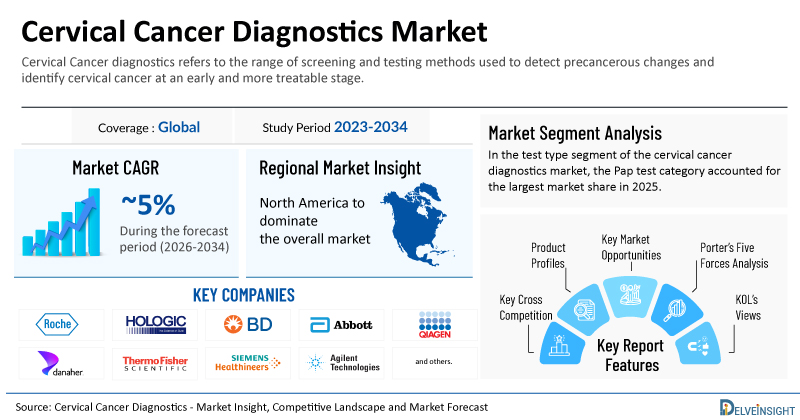
- 2025 Cervical Cancer Diagnostics Market Size: USD 4.4 Billion
- 2034 Projected Cervical Cancer Diagnostics Market Size: USD 7.1 Billion
- Cervical Cancer Diagnostics Market Growth Rate (2026-2034): ~5%
- Largest Cervical Cancer Diagnostics Market: North America
- Largest Test Type Segment: Pap Test Category
- Key Companies in the Cervical Cancer Diagnostics Market: F. Hoffmann-La Roche Ltd, Hologic, Inc., Becton, Dickinson and Company, Abbott Laboratories, Qiagen N.V., Danaher Corporation, Thermo Fisher Scientific, Siemens Healthineers, Agilent Technologies, bioMérieux SA, Bio-Rad Laboratories, Illumina, Inc., Myriad Genetics, PerkinElmer, Inc., Sysmex Corporation, Olympus Corporation, Carl Zeiss Meditec AG, Leisegang GmbH, CooperSurgical Inc., MobileODT Ltd., and others
To read more about the latest highlights related to the cervical cancer diagnostics market, get a snapshot of the key highlights @ https://www.delveinsight.com/sample-request/cervical-cancer-diagnostics-market
Key Factors Contributing to the Rise in Growth of the Cervical Cancer Diagnostics Market
- Increasing Prevalence of Cervical Cancer and HPV Infections: The rising incidence of cervical cancer, primarily caused by persistent HPV infections, is significantly driving the demand for advanced diagnostic solutions. Growing awareness about the importance of early detection is encouraging more women to undergo regular screening tests.
- Growing Adoption of HPV Testing and Molecular Diagnostics: Healthcare providers are increasingly shifting toward HPV DNA testing, biomarker-based screening, and molecular diagnostic technologies due to their higher accuracy and sensitivity compared to conventional Pap smear tests. These advanced techniques enable earlier and more reliable detection of precancerous lesions.
- Government Screening Programs and Awareness Campaigns: Several governments and healthcare organizations are implementing nationwide cervical cancer screening initiatives and awareness campaigns to promote routine testing. Public health programs aimed at improving women’s healthcare access are boosting diagnostic testing volumes globally.
- Technological Advancements in Diagnostic Platforms: Continuous innovations in diagnostic technologies, including AI-enabled imaging systems, liquid-based cytology, and automated screening platforms, are improving testing efficiency and reducing diagnostic errors. These advancements are enhancing the overall adoption of cervical cancer diagnostics.
- Rising Healthcare Expenditure and Improved Healthcare Infrastructure: Increasing investments in healthcare infrastructure, particularly in emerging economies, are expanding access to cancer screening services. The availability of modern laboratories and diagnostic centers is contributing to market growth.
- Growing Focus on Preventive Healthcare: The global shift toward preventive healthcare and early disease detection is encouraging routine cervical cancer screening among women. Increasing health consciousness and regular gynecological check-ups are supporting market expansion.
- Expansion of Diagnostic Laboratories and Point-of-Care Testing: The rapid growth of diagnostic laboratories, along with the emergence of point-of-care and portable testing solutions, is improving accessibility to cervical cancer diagnostics, especially in remote and underserved regions.
- Supportive Reimbursement Policies and Insurance Coverage: Favorable reimbursement frameworks and wider insurance coverage for cervical cancer screening procedures in developed countries are encouraging higher patient participation in diagnostic testing programs.
- Growing Geriatric and High-Risk Female Population: The increasing population of women aged 30 years and above, who are more susceptible to HPV-related cervical abnormalities, is creating sustained demand for regular cervical cancer screening and diagnostic services.
Get a sneak peek at the cervical cancer diagnostics market dynamics @ Cervical Cancer Diagnostics Market Trends
Regional Cervical Cancer Diagnostics Market Insights
North America
- North America held the largest share of the cervical cancer diagnostics market, accounting for nearly 44% in 2025.
- The region’s dominance can be attributed to its advanced healthcare infrastructure, widespread adoption of HPV testing and molecular diagnostic technologies, and growing awareness of the importance of early cancer detection.
- Established screening initiatives backed by organizations such as the Centers for Disease Control and Prevention and the American Cancer Society continue to encourage routine Pap smear and HPV screening across the population.
- Moreover, the strong presence of leading industry participants, ongoing product innovations, and supportive reimbursement frameworks are expected to strengthen the region’s market leadership further.
Europe
- The European cervical cancer diagnostics market is experiencing steady and robust expansion, driven by the growing incidence of cervical cancer and the increasing focus on organized screening initiatives throughout the region.
- Cervical cancer continues to pose a major public health challenge, with a substantial number of new cases diagnosed each year, largely linked to Human papillomavirus infections, thereby increasing the demand for timely and routine screening.
- In addition, governments and healthcare organizations across Europe are strengthening structured screening efforts through initiatives such as Europe’s Beating Cancer Plan, which aims to enhance screening accessibility and support earlier detection.
- Moreover, evolving clinical recommendations are increasingly favoring HPV-based primary screening over conventional cytology because of its superior sensitivity and diagnostic accuracy, further supporting market growth and adoption.
Asia-Pacific
- The Asia Pacific region is becoming a key contributor to the growth of the cervical cancer diagnostics market, supported by the increasing prevalence of the disease, advancements in healthcare infrastructure, and expanding screening efforts.
- The region represents a considerable portion of global cervical cancer cases, primarily due to the rising incidence of Human papillomavirus infections, which is driving demand for early diagnosis and regular screening programs.
- In addition, rapid urban development and growing healthcare expenditure across countries such as China, India, and Southeast Asian nations are improving access to diagnostic centers and laboratory services.
- Governments throughout the region are also strengthening awareness campaigns and launching nationwide screening initiatives, leading to higher testing rates.
- The increasing adoption of innovative technologies, such as molecular HPV diagnostics and self-collection screening methods, is further supporting market expansion.
- At the same time, the availability of affordable diagnostic solutions designed for cost-conscious markets is enhancing accessibility and encouraging wider adoption.
- Consequently, the Asia Pacific cervical cancer diagnostics market is expected to witness substantial growth, contributing significantly to the expansion of the global market.
To know more about why North America is leading the market growth in the cervical cancer diagnostics market, get a snapshot of the Cervical Cancer Diagnostics Market Share
Recent Developmental Activities in the Cervical Cancer Diagnostics Market
- In April 2026, Waters Corporation announced that the U.S. Food and Drug Administration (FDA) had cleared the Onclarity HPV Self-Collection Kit and approved the BD Onclarity HPV Assay with extended genotyping for at-home use, marking a significant milestone in expanding access to cervical cancer screening and removing barriers that currently prevent many individuals from receiving routine screening.
- In February 2026, Hologic, Inc. announced that its Aptima® HPV Assay received FDA approval for clinician-collected HPV primary screening. Hologic’s human papillomavirus (HPV) test is the only FDA-approved mRNA-based assay, designed specifically to detect infections most likely to lead to cervical cancer.
- In January 2026, Teal Health, a virtual women’s health company on a mission to eliminate cervical cancer in the U.S., announced the national availability of its at-home cervical cancer screening (PAP Test), which included the FDA-authorized Teal WandTM self-collection device and comprehensive telehealth platform. Cervical cancer screening, often referred to as the Pap smear, is a recommended screening for women ages 25-65.
What are Cervical Cancer Diagnostics?
Cervical Cancer diagnostics refers to the range of screening and testing methods used to detect precancerous changes and identify cervical cancer at an early and more treatable stage. These diagnostic approaches primarily include Pap smear tests, HPV testing, colposcopy, biopsy procedures, and advanced molecular diagnostic techniques. Routine screening helps identify abnormal cervical cells before they develop into invasive cancer, significantly reducing mortality rates. In recent years, the field of cervical cancer diagnostics has evolved with the introduction of liquid-based cytology, biomarker-based assays, and AI-enabled screening technologies, improving accuracy, sensitivity, and accessibility. Increasing awareness programs, government-led screening initiatives, and growing adoption of preventive healthcare practices are further driving the demand for cervical cancer diagnostic solutions worldwide.
| Cervical Cancer Diagnostics Market Report Metrics | Details |
| Coverage | Global |
| Study Period | 2023–2034 |
| Cervical Cancer Diagnostics Market CAGR | ~5% |
| Cervical Cancer Diagnostics Market Size by 2034 | USD 7.1 Billion |
| Key Cervical Cancer Diagnostics Companies | F. Hoffmann-La Roche Ltd, Hologic, Inc., Becton, Dickinson and Company, Abbott Laboratories, Qiagen N.V., Danaher Corporation, Thermo Fisher Scientific, Siemens Healthineers, Agilent Technologies, bioMérieux SA, Bio-Rad Laboratories, Illumina, Inc., Myriad Genetics, PerkinElmer, Inc., Sysmex Corporation, Olympus Corporation, Carl Zeiss Meditec AG, Leisegang GmbH, CooperSurgical Inc., MobileODT Ltd., and others |
Cervical Cancer Diagnostics Market Assessment
- Cervical Cancer Diagnostics Market Segmentation
- Cervical Cancer Diagnostics Market Segmentation By Test Type: Pap Test, HPV Testing, Colposcopy, Cervical Biopsy, and Others
- Cervical Cancer Diagnostics Market Segmentation By Product Type: Instruments, Consumables, and Software
- Cervical Cancer Diagnostics Market Segmentation By Age Group: 20-40 Years and Above 40 Years
- Cervical Cancer Diagnostics Market Segmentation By End User: Hospitals, Diagnostic Centers, and Others
- Cervical Cancer Diagnostics Market Segmentation By Geography: North America, Europe, Asia-Pacific, and Rest of World
- Porter’s Five Forces Analysis, Product Profiles, Case Studies, KOL’s Views, Analyst’s View
Which MedTech key players in the cervical cancer diagnostics market are set to emerge as the trendsetter explore @ Cervical Cancer Diagnostics Market Analysis
Table of Contents
| 1 | Cervical Cancer Diagnostics Market Report Introduction |
| 2 | Cervical Cancer Diagnostics Market Executive Summary |
| 3 | Cervical Cancer Diagnostics Market Key Factors Analysis |
| 4 | Impact Analysis |
| 5 | Regulatory Analysis |
| 6 | Cervical Cancer Diagnostics Market Porter’s Five Forces Analysis |
| 7 | Cervical Cancer Diagnostics Market Assessment |
| 8 | Competitive Landscape |
| 9 | Startup Funding & Investment Trends |
| 10 | Cervical Cancer Diagnostics Market Company and Product Profiles |
| 11 | KOL Views |
| 12 | Project Approach |
| 13 | About DelveInsight |
| 14 | Disclaimer & Contact Us |
Interested in knowing the cervical cancer diagnostics market share by 2034? Click to get a snapshot of the Cervical Cancer Diagnostics Market Size
Related Reports
Cervical Cancer Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key cervical cancer companies, including Roche, AstraZeneca, Precigen, Puma Biotechnology, Iovance Biotherapeutics, Nykode Therapeutics, ISA Pharmaceuticals, Regeneron, Transgene, Daiichi Sankyo, and others.
Cervical Cancer Clinical Trial Analysis
Cervical Cancer Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key cervical cancer companies, including Regeneron Pharmaceuticals, Jiangsu Hengrui Medicine, Akeso Biopharma, Celgene, Henlix Biotech, Zeria, Advaxis, Akeso Biopharma, AnGes, Advenchen Laboratories, Jiangsu Chia Tai Tianqing Pharmaceutical, Valent recombinant vaccine – National Vaccine and Serum Institute, Xiamen Innovax Biotech, Genentech, Celgene, MedImmune, Bristol-Myers Squibb, Ono Pharmaceuticals, InnoMab, BeiGene, Agenus, Ludwig Institute for Cancer Research, EMD Serono, Merck, Apollomics, Immunitor, Genentech, Arcus Biosciences, Guangzhou Gloria Biosciences, Taiho Pharmaceutical, Innovent Biologics, Clovis Oncology, Janssen, GlaxoSmithKline, Iovance Biotherapeutics, and others.
HPV Testing and Pap Test Market
HPV Testing and Pap Test Market Insights, Competitive Landscape, and Market Forecast – 2034 report deliver an in-depth understanding of the market trends, market drivers, market barriers, and key HPV testing and Pap test companies, including F. Hoffmann-La Roche, Hologic Inc., Becton Dickinson, QIAGEN, Abbott Laboratories, Danaher Corporation, Siemens Healthineers, Seegene Inc., Bio-Rad Laboratories, Thermo Fisher Scientific, Femasys Inc., Arbor Vita Corporation, Quest Diagnostics, LabCorp, Sysmex Corporation, FUJIFILM Holdings, Olympus Corporation, CooperSurgical, Cardinal Health, Nihon Kohden, and others.
HPV Vaccines Market Insight, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of market trends, market drivers, market barriers, and key HPV vaccines companies, including GlaxoSmithKline, Xiamen Innovax, Walvax Biotechnology, Merck & Co., Serum Institute of India, and others.
AI in Cancer Diagnostics Market
AI in Cancer Diagnostics Market Insights, Competitive Landscape, and Market Forecast – 2034 report deliver an in-depth understanding of the market trends, market drivers, market barriers, and key AI in cancer diagnostics companies, including iCAD, Inc., ibex-ai, Roche Diagnostics, Kheiron Medical Technologies Limited, MVision AI Inc., Siemens Healthineers AG, GE HealthCare, NVIDIA Corporation, Digital Diagnostics Inc., IBM Corporation, Azra AI, ConcertAI, PathAI, Median Technologies, Paige AI Inc., Therapixel, Flatiron, Freenome Holdings Inc., Onc.AI, Sonrai Analytics, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur [email protected] +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaChron.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaChron.com takes no editorial responsibility for the same.