The non-small cell lung cancer clinical trial analysis report delivers important insights into ongoing research of 120+ pipeline non-small cell lung cancer drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
New York, USA, April 16, 2026 (GLOBE NEWSWIRE) — Non-Small Cell Lung Cancer Clinical Trial Race Intensifies as 100+ Companies Competing in Therapeutic Segment Worldwide | DelveInsight
The non-small cell lung cancer clinical trial analysis report delivers important insights into ongoing research of 120+ pipeline non-small cell lung cancer drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
DelveInsight’s ‘Non-Small Cell Lung Cancer Pipeline Insight 2026’ report provides comprehensive global coverage of pipeline therapies for non-small cell lung cancer across various stages of clinical development. The report offers an in-depth analysis of key trends, emerging therapies, and competitive landscape dynamics, highlighting the strategies of major pharmaceutical companies to advance the pipeline and capitalize on future growth opportunities. In addition, it includes critical insights into clinical trial benchmarking, partnering and licensing activities, and regulatory pathways involving the FDA and EMA, enabling stakeholders to make informed decisions and optimize development strategies within the non-small cell lung cancer domain.
Non-Small Cell Lung Cancer Clinical Trial Analysis Summary
- DelveInsight’s non-small cell lung cancer pipeline report depicts a robust space with 100+ active players working to develop 120+ pipeline non-small cell lung cancer drugs.
- Key non-small cell lung cancer companies, such as MAIA Biotechnology, SystImmune, Nuvalent, Pfizer, AbbVie, Revolution Medicines, GlaxoSmithKline, Compugen, BioNTech, Candel Therapeutics, OncoC4, BioNTech, Incyte, Macrogenics, Iovance Biotherapeutics, Hoffmann-La Roche, Genentech, Regeneron Pharmaceuticals, Merck, Kelun-Biotech, Immutep, Moderna Therapeutics, BeyondSpring, Cullinan Therapeutics, Eli Lilly and Company, AstraZeneca, OSE Immunotherapeutics, ImmunityBio, Jiangsu HengRui Medicine, Gilead Sciences, Arcus Biosciences, Merck, Otsuka Pharmaceutical (Taiho and Astex Pharmaceuticals), ArriVent BioPharma, Shanghai Allist Pharmaceuticals, Innovent Biologics, Takeda Pharmaceutical, Akeso Bio, Summit Therapeutics, Apollomics, Avistone Biotechnology, and others are evaluating new non-small cell lung cancer drugs to improve the treatment landscape.
- Promising pipeline non-small cell lung cancer therapies, such as Ateganosine, BMS-986504 + Pembrolizumab, Iza-bren (izalontamab brengitecan), Neladalkib (NVL-655), PF-08046054 (SGN-PDL1V), Telisotuzumab adizutecan, Temab-A (ABBV-400), Daraxonrasib (RMC-6236), ZEJULA (niraparib), Rilvegostomig (AZD2936), Pumitamig (BNT327/PM8002), CAN-2409 (aglatimagene besadenovec), Gotistobart (BNT316), ZYNYZ (retifanlimab-dlwr), LN-145, Divarasib (GDC-6036), Fianlimab (REGN3767), Sacituzumab Tirumotecan (MK-2870), Eftilagimod alpha (Efti, IMP321), Intismeran autogene (mRNA-4157/V940), Plinabulin, Zipalertinib (CLN-081), Olomorasib (LY3537982), Ceralasertib (AZD6738), TEDOPI (OSE2101), Sigvotatug Vedotin (PF08046047, SGN-B6A), ANKTIVA (N-803, nogapendekin alfa inbakicept-pmln), Pyrotinib, Volrustomig (MEDI5752), TRODELVY (sacituzumab govitecan), Domvanalimab + Combination Therapies, OPDUALAG (nivolumab and relatlimab), Calderasib (MK-1084), Firmonertinib, Livmoniplimab (ARGX-115), IBI363, Ivonescimab (AK112), Vebreltinib (APL-101), Zidesamtinib (NVL-520), and others, are in different phases of non-small cell lung cancer clinical trials.
- Approximately 30+ non-small cell lung cancer drugs are in the late stages of development.
- Notable MoAs in non-small cell lung cancer clinical trials include PRMT5 inhibitor + PD-1 inhibitor, EGFR × HER3 inhibitor + EGFR TKI, ALK inhibitor, PD-L1 inhibitor, DNA synthesis inhibitors, Immunostimulants, Gene transferase, EGFR antagonist, CD3 inhibitor, LLT1 antagonist, T lymphocyte replacements, and others.
Request a sample and discover the recent advances in non-small cell lung cancer drugs development @ https://www.delveinsight.com/sample-request/non-small-cell-lung-cancer-pipeline-insight
What is Non-Small Cell Lung Cancer?
Non-small cell lung cancer (NSCLC) is the most common type of lung cancer, accounting for roughly 85% of all cases. It is a group of lung cancers that behave and respond to treatment in similar ways, with the main subtypes being adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. NSCLC typically develops more slowly than small-cell lung cancer, but it is often diagnosed at an advanced stage due to subtle or absent early symptoms. Major risk factors include smoking, exposure to secondhand smoke, air pollution, and certain occupational hazards. Advances in understanding tumor biology, especially genetic mutations such as EGFR and ALK, have led to more targeted therapies and immunotherapies, significantly improving outcomes for some patients.
Find out more about non-small cell lung cancer drugs @ Non-Small Cell Lung Cancer Treatment
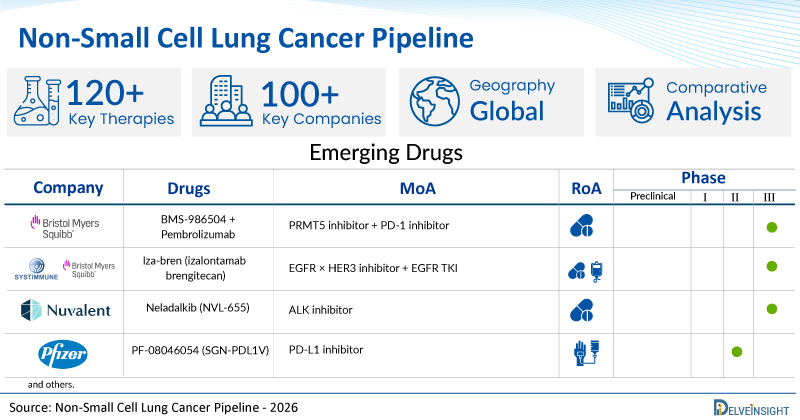
A snapshot of the Pipeline Non-Small Cell Lung Cancer Drugs mentioned in the report:
| Drug | Company | Phase | MoA | RoA |
| BMS-986504 + Pembrolizumab | BMS | II/III | PRMT5 inhibitor + PD-1 inhibitor | Oral |
| Iza-bren (izalontamab brengitecan) | SystImmune and Bristol Myers Squibb | III | EGFR × HER3 inhibitor + EGFR TKI | Oral + IV |
| Neladalkib
(NVL-655) |
Nuvalent | III | ALK inhibitor | Oral |
| PF-08046054
(SGN-PDL1V) |
Pfizer | III | PD-L1 inhibitor | Intravenous infusion |
| FF-10832 | FUJIFILM Corporation | II | DNA synthesis inhibitors | Intravenous |
| BNT116 | BioNTech SE/Regeneron Pharmaceuticals | II | Immunostimulants | Intravenous |
| CAN-2409 | Candel Therapeutics | II | Gene transferase | Intravenous |
| JANX008 | Janux Therapeutics | I | EGFR antagonist; CD3 inhibitor | Intravenous |
| ZM-008 | Zumutor Biologics | I | LLT1 antagonist | Intravenous |
| ADP-600 | Adaptimmune | Preclinical | T lymphocyte replacements | Oral |
Learn more about the emerging non-small cell lung cancer therapies @ Non-Small Cell Lung Cancer Clinical Trials
Stuti Mahajan, consulting manager at DelveInsight, said that the emerging NSCLC therapies are increasingly driven by precision oncology, with next-generation targeted agents (e.g., ROS1, HER2, KRAS inhibitors) and bispecific/immunotherapy combinations showing strong clinical promise and differentiation. Recent approvals and pipeline data suggest improved efficacy and durability, particularly in biomarker-selected populations, indicating a shift toward more personalized treatment paradigms. She further added, NSCLC pipeline looks highly competitive but innovation-rich, with antibody-drug conjugates and novel kinase inhibitors expected to drive the next wave of growth and address unmet needs in resistant or hard-to-treat subtypes.
Recent Developments in Non-Small Cell Lung Cancer Treatment Space
- In March 2026, TRIANA Biomedicines, Inc. announced that the FDA had granted fast track designation to TRI-611, an investigational molecular glue degrader therapy, as a treatment for those with ALK-positive non–small cell lung cancer (NSCLC).
- In March 2026, Eisai Co., Ltd. and Nuvation Bio Inc. announced that the European Medicines Agency (EMA) had validated the Marketing Authorisation Application (MAA) for taletrectinib for the treatment of advanced ROS1-positive (ROS1+) non-small cell lung cancer (NSCLC). The filing will follow a standard review timeline.
- In March 2026, Researchers reported at the 2026 European Lung Cancer Congress that Hernexeos (zongertinib) led to high response rates and disease control in patients with advanced HER2-mutant non–small cell lung cancer (NSCLC), including those with active brain metastases, addressing an unmet need for targeted first-line treatment options in this population.
- In February 2026, the U.S. Food and Drug Administration (FDA) granted Regenerative Medicine Advanced Therapy (RMAT) designation to KB707, an investigational inhaled immunotherapy being developed by Krystal Biotech for the treatment of advanced or metastatic non-small cell lung cancer (NSCLC).
- In February 2026, the FDA received a Biologics License Application (BLA) for ivonescimab in combination with chemotherapy for the treatment of advanced or metastatic EGFR-mutated non-small cell lung cancer (NSCLC).
- In February 2026, the FDA approved the subcutaneous formulation of amivantamab (RYBREVANT) for patients with EGFR-mutated NSCLC. This new delivery method, combined with recombinant human hyaluronidase, significantly reduces administration time from several hours to approximately five minutes.
- In February 2026, Johnson & Johnson confirmed the FDA approval of RYBREVANT FASPPRO (amivantamab-vmjw and hyaluronidase-zyjk). This subcutaneous injection is approved for use as a monotherapy or in combination regimens across multiple EGFR-mutated lung cancer indications.
- In January 2026, AbbVie and RemeGen announced an exclusive licensing agreement for the development, manufacturing, and commercialization of RC148, a novel investigational Programmed Cell Death-1 (PD-1)/Vascular Endothelial Growth Factor (VEGF)-targeted bispecific antibody.
- In January 2026, the US FDA accepted for filing Summit’s Biologics License Application (BLA) for the HARMONi Phase III trial and assigned a Prescription Drug User Fee Act (PDUFA) goal action date of November 14, 2026.
- In January 2026, the FDA granted orphan drug designation to gotistobart (BNT316/ONC-392) for the treatment of patients with squamous non–small cell lung cancer (NSCLC). The announcement, made by BioNTech SE and OncoC4, Inc, highlights the investigational anti–CTLA-4 monoclonal antibody’s potential in treating an aggressive subset of lung cancer that has historically seen limited therapeutic breakthroughs following the failure of first-line immunotherapy.
- In November 2025, Nuvalent reported positive topline pivotal data from its ALKOVE-1 trial of neladalkib for patients with advanced ALK-positive NSCLC who were previously treated with TKIs. The drug showed a high objective response rate (ORR) and significant intracranial activity, addressing a major unmet need for patients with brain metastases.
- In November 2025, a New Drug Application (NDA) was submitted for zipalertinib for the treatment of EGFR exon 20–mutant NSCLC, with completion and priority review planned for Q1 2026, based on data from the REZILIENT1 Phase I/II study.
- In October 2025, Pfizer announced updated Phase 2 PHAROS trial results showing sustained long-term survival for BRAFTOVI (encorafenib) and MEKTOVI (binimetinib) in patients with BRAF V600E-mutant metastatic non-small cell lung cancer (NSCLC).
- In October 2025, collaborators from Genprex reported positive preclinical data for REQORSA gene therapy at the 2025 AACR-NCI-EORTC conference. The findings showed that REQORSA, when combined with pembrolizumab (KEYTRUDA), significantly inhibited tumor growth in models of KRAS-mutant lung cancer.
- In October 2025, OncoC4 announced that China’s National Medical Product Administration (NMPA) had granted Breakthrough Therapy Designation (BTD) for gotistobart (BNT316/ONC392) for the treatment of patients with squamous non-small cell lung cancer (sqNSCLC) who have progressed on prior standard immuno-oncology therapies (IO).
Scope of the Non-Small Cell Lung Cancer Pipeline Report
- Coverage: Global
- Non-Small Cell Lung Cancer Therapeutic Assessment By Product Type: Mono, Combination, Mono/Combination
- Non-Small Cell Lung Cancer Therapeutic Assessment By Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
- Non-Small Cell Lung Cancer Therapeutics Assessment By Route of Administration: Oral, Intravenous, Subcutaneous, Parenteral, Topical
- Non-Small Cell Lung Cancer Therapeutics Assessment By Molecule Type: Recombinant fusion proteins, Small molecule, Monoclonal antibody, Peptide, Polymer, Gene therapy
- Non-Small Cell Lung Cancer Therapeutics Assessment By Mechanism of Action: PRMT5 inhibitor + PD-1 inhibitor, EGFR × HER3 inhibitor + EGFR TKI, ALK inhibitor, PD-L1 inhibitor, DNA synthesis inhibitors, Immunostimulants, Gene transferase, EGFR antagonist, CD3 inhibitor, LLT1 antagonist, T lymphocyte replacements, and others
- Key Non-Small Cell Lung Cancer Companies: MAIA Biotechnology, SystImmune, Nuvalent, Pfizer, AbbVie, Revolution Medicines, GlaxoSmithKline, Compugen, BioNTech, Candel Therapeutics, OncoC4, BioNTech, Incyte, Macrogenics, Iovance Biotherapeutics, Hoffmann-La Roche, Genentech, Regeneron Pharmaceuticals, Merck, Kelun-Biotech, Immutep, Moderna Therapeutics, BeyondSpring, Cullinan Therapeutics, Eli Lilly and Company, AstraZeneca, OSE Immunotherapeutics, ImmunityBio, Jiangsu HengRui Medicine, Gilead Sciences, Arcus Biosciences, Merck, Otsuka Pharmaceutical (Taiho and Astex Pharmaceuticals), ArriVent BioPharma, Shanghai Allist Pharmaceuticals, Innovent Biologics, Takeda Pharmaceutical, Akeso Bio, Summit Therapeutics, Apollomics, Avistone Biotechnology, and others.
- Key Non-Small Cell Lung Cancer Pipeline Therapies: Ateganosine, BMS-986504 + Pembrolizumab, Iza-bren (izalontamab brengitecan), Neladalkib (NVL-655), PF-08046054 (SGN-PDL1V), Telisotuzumab adizutecan, Temab-A (ABBV-400), Daraxonrasib (RMC-6236), ZEJULA (niraparib), Rilvegostomig (AZD2936), Pumitamig (BNT327/PM8002), CAN-2409 (aglatimagene besadenovec), Gotistobart (BNT316), ZYNYZ (retifanlimab-dlwr), LN-145, Divarasib (GDC-6036), Fianlimab (REGN3767), Sacituzumab Tirumotecan (MK-2870), Eftilagimod alpha (Efti, IMP321), Intismeran autogene (mRNA-4157/V940), Plinabulin, Zipalertinib (CLN-081), Olomorasib (LY3537982), Ceralasertib (AZD6738), TEDOPI (OSE2101), Sigvotatug Vedotin (PF08046047, SGN-B6A), ANKTIVA (N-803, nogapendekin alfa inbakicept-pmln), Pyrotinib, Volrustomig (MEDI5752), TRODELVY (sacituzumab govitecan), Domvanalimab + Combination Therapies, OPDUALAG (nivolumab and relatlimab), Calderasib (MK-1084), Firmonertinib, Livmoniplimab (ARGX-115), IBI363, Ivonescimab (AK112), Vebreltinib (APL-101), Zidesamtinib (NVL-520), and others.
Dive deep into rich insights for new non-small cell lung cancer treatments, visit @ Non-Small Cell Lung Cancer Drugs
Table of Contents
| 1. | Non-Small Cell Lung Cancer Pipeline Report Introduction |
| 2. | Non-Small Cell Lung Cancer Pipeline Report Executive Summary |
| 3. | Non-Small Cell Lung Cancer Pipeline: Overview |
| 4. | Analytical Perspective In-depth Commercial Assessment |
| 5. | Non-Small Cell Lung Cancer Clinical Trial Therapeutics |
| 6. | Non-Small Cell Lung Cancer Pipeline: Late-Stage Products (Pre-registration) |
| 7. | Non-Small Cell Lung Cancer Pipeline: Late-Stage Products (Phase III) |
| 8. | Non-Small Cell Lung Cancer Pipeline: Mid-Stage Products (Phase II) |
| 9. | Non-Small Cell Lung Cancer Pipeline: Early-Stage Products (Phase I) |
| 10. | Non-Small Cell Lung Cancer Pipeline Therapeutics Assessment |
| 11. | Inactive Products in the Non-Small Cell Lung Cancer Pipeline |
| 12. | Company-University Collaborations (Licensing/Partnering) Analysis |
| 13. | Key Companies |
| 14. | Key Products in the Non-Small Cell Lung Cancer Pipeline |
| 15. | Unmet Needs |
| 16. | Market Drivers and Barriers |
| 17. | Future Perspectives and Conclusion |
| 18. | Analyst Views |
| 19. | Appendix |
For further information on the non-small cell lung cancer cure research, reach out @ Medication for Non-Small Cell Lung Cancer Treatment
Related Reports
Non-Small Cell Lung Cancer Market
Non-Small Cell Lung Cancer Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key NSCLC companies including BeiGene, Merus and Partner Therapeutics, Johnson & Johnson Innovative Medicine, Betta Pharma/Xcovery, CStone Pharmaceuticals, Bristol-Myers Squibb, Haihe Biopharma, Taiho Pharmaceutical, Takeda Pharmaceuticals, EMD Serono (Merck KGaA), Eli Lilly and Company, Merck, Regeneron/Sanofi, Ono Pharmaceutical, Novartis, Genentech/Roche, Amgen, Chugai Laboratories, Moderna Therapeutics, Pfizer, GSK, and others.
Small Cell Lung Cancer Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key advanced small cell lung cancer companies, including Hoffmann-La Roche, Pharma Mar, Jazz, Daiichi Sankyo, Henlius Biotech, SN BioScience, Merck & Co, AstraZeneca, Advenchen Laboratories, and others.
Small-cell Lung Cancer Clinical Trial Analysis
Small-cell Lung Cancer Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key small-cell lung cancer companies, including Ascentage Pharma, Merck & Co, AstraZeneca, Advenchen Laboratories, GlaxoSmithKline, Advanced Accelerator Applications, Trillium Therapeutics, Vernalis, Oncoceutics, NewBio Therapeutics, Wigen Biomedicine, Linton Pharm, Carrick Therapeutics, Xencor, Jiangsu HengRui Medicine, Aileron Therapeutics, Roche, Ipsen, Celgene, Lee’s Pharmaceutical Limited, AbbVie, G1 Therapeutics, Chipscreen Biosciences, Luye Pharma Group, Shanghai Henlius Biotech, CSPC ZhongQi Pharmaceutical Technology, Impact Therapeutics, and others.
EGFR NSCLC Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key EGFR NSCLC companies including ArriVent BioPharma, Akeso Biopharma, Summit Therapeutics, J INTS BIO, Merus, Merck, Kelun-Biotech, Genprex, Cullinan Oncology, Taiho Pharma, CSPC Pharmaceutical, AbbVie, Teligene, Bristol–Myers Squibb, SystImmune, Black Diamond Therapeutics, Lantern Pharma, Dizal Pharmaceutical, Daiichi Sankyo, AstraZeneca, Jiangsu Hansoh Pharmaceutical, Boehringer Ingelheim, Pfizer, Johnson & Johnson Innovative Medicine, Yuhan Corporation, Eli Lilly and Company, and others.
EGFR-NSCLC Clinical Trial Analysis
EGFR-NSCLC Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key EGFR-NSCLC companies, including Genprex, J INTS Bio, Black Diamond Therapeutics, BlossomHill Therapeutics, Scorpion Therapeutics, Pierre Fabre, Antares Therapeutics, Bayer, A2 Biotherapeutics, BioNTech, Dragonfly Therapeutics, Merus, Taiho Oncology, BeOne Medicines (formerly BeiGene), Ono Pharmaceutical, Avistone Biotechnology, Dizal Pharmaceuticals, Checkpoint Therapeutics, DEKA Biosciences, Taiho Oncology, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur [email protected] +14699457679
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaChron.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaChron.com takes no editorial responsibility for the same.